

Allston: Imaging & Analysis
Our Allston SEC core laboratory spaces provide equipment and staff expertise in materials characterization, electron microscopy and soft lithography.

The Center for Nanoscale Systems provides three facilities at the Science and Engineering Complex (SEC) at Harvard’s Allston campus. Our new materials characterization lab houses a diverse selection of instrumentation for analyzing hard and soft materials, including thermal, mechanical, surface, particle, and chemical characterization. Meanwhile, the new imaging lab has two high resolution electron microscopes, along with an array of electron microscopy sample preparation tools for hard, soft and biological materials.

Atomic Force Microscopy
Atomic Force Microscopy (AFM) can be used to determine the surface topography in air or liquids with atomic height resolution. It can be used to perform stiffness mapping with Quantitative Imaging force curves, in addition to “microrheology” on viscoelastic materials. An inverted optical microscope can align surface scans with fluorescent imaging. Heated stages are available for both solids and liquids.
More information on the JPK Nanowizard AFM.
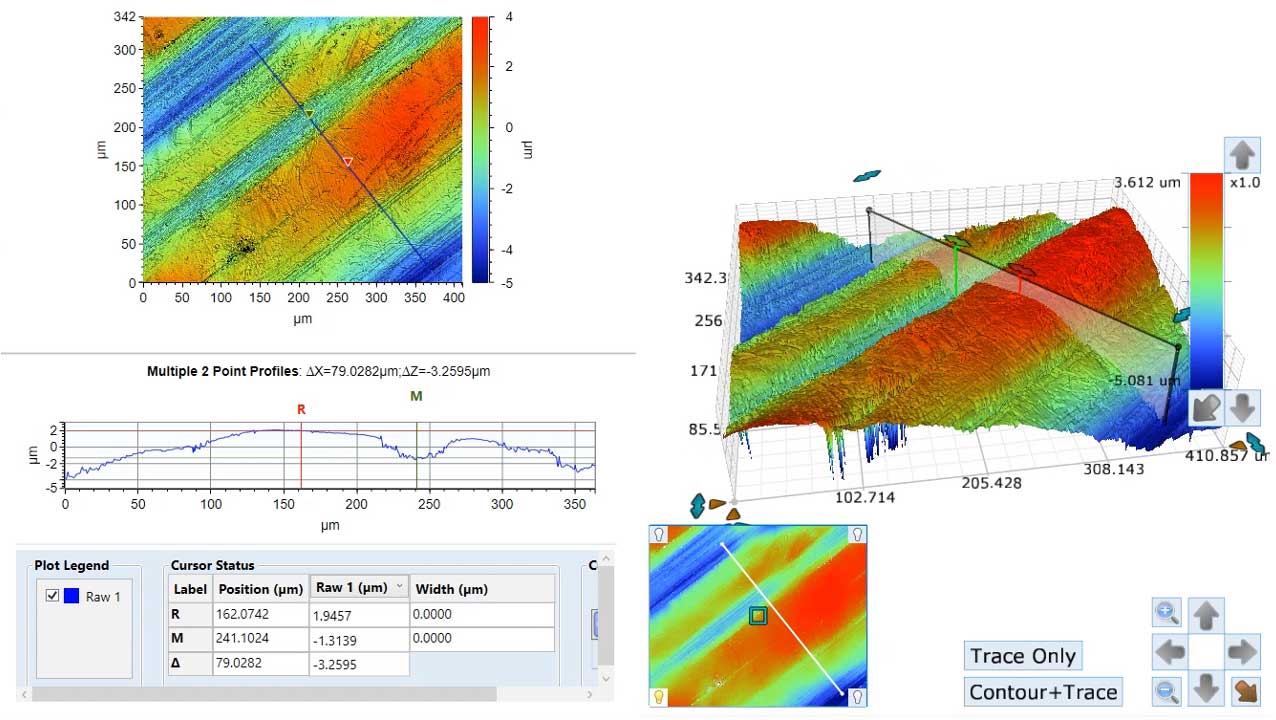
Optical Profilometry
Optical profilometry can be used to quickly and easily measure surface topography over large areas. The interferometric optical profiler enables the collection of quantitative surface topographic images with nanometer z and micron x-y spatial resolution.
More information on our Bruker Optical Interferometric Profiler.

Electrical Characterization
Our probe station can be used to measure the electrical properties of devices.
More information on our Signatone Probe Station.

Dynamic Mechanical Analysis
The Dynamic Mechanical Analyzer (DMA) can be used to determine the mechanical properties of viscoelastic materials such as polymers and other solids. It can be operated in bending, tension, shear and compression modes. Specimens can be studied over a wide range of temperatures (-190 °C to 600 °C) and frequencies (0.001 Hz to 300 Hz).
More information on our Mettler Toledo DMA 1.

Rheometry
Rheometry is used to evaluate the viscoelastic/rheological properties of solids and liquids. Beyond analyzing materials over a wide range of temperatures, -160 to 600 C with the Environmental Test Chamber, an ultraviolet accessory is available to characterize UV curing or degradation, and the dielectric properties of materials can be analyzed with the LCR meter accessory.
More information on our TA Instruments HR 20 Discovery Hybrid Rheometer.

Nanoindentation
The nanoindenter can quickly and easily probe the modulus and hardness of materials. This technique is also known as depth sensing indentation or instrumented indentation. It can perform dynamic analyses to determine viscoelastic properties.
More information on our Bruker Nanoindenter.

Thermogravimetric Analysis
Thermogravimetric analysis (TGA) can quantify thermal decomposition under nitrogen or air, at temperatures of up to 1000 °C. It can be used to analyze gas phase decomposition products when used with our Fourier transform infrared (FTIR) accessory. The system can determine precise compositions of organic/inorganic hybrid systems, or can be used to calculate the moisture content of hydrogels and other solids.
More information on our TA Instruments Discovery TGA 550.
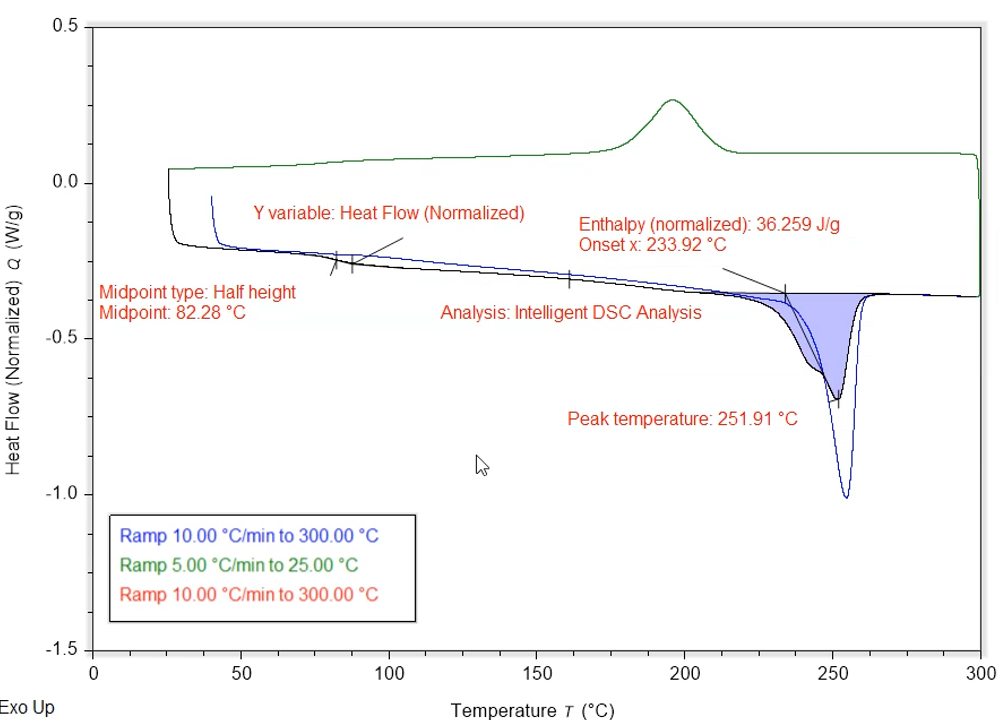
Differential Scanning Calorimetry
For purely thermal analysis, Differential Scanning Calorimetry (DSC) is used to quantify the melting, crystallization, and glass transitions between -90 °C and 550 °C. Modulated DSC can be used to distinguish between kinetic and thermodynamic heatflows.
More information on our TA Instruments Discovery 250 DSC.

Raman Spectroscopy
Raman spectroscopy facilitates the study of vibrational and other low frequency optical modes in a specimen. When combined with a motorized stage, such systems can be used to gain chemical information with sub-micron spatial resolution. The Raman system at the SEC has 532 nm and 325 nm laser lines available.
More information on our Horiba LabRam Soleil Raman Microscope.
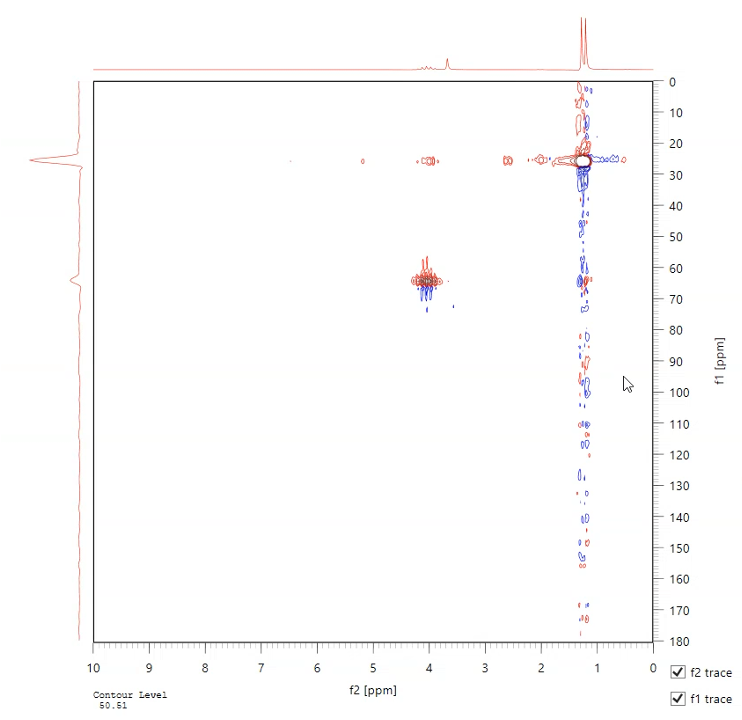
NMR Spectroscopy
Nuclear magnetic resonance (NMR) can be used to determine molecular composition. A magnetic field is applied to the specimen and radio frequency pulses are used to measure the resonant frequency of atomic nucleus. The benchtop system at the SEC uses 1H, 13C and 19F isotopes. Two dimensional analysis modes include correlated spectroscopy (COSY), rotating frame Overhause effect spectroscopy (ROESY), heteronuclear multiple quantum coherence (HMQC) and heteronuclear single quantum coherence (HSQC) modes.
More information on our Magritek Spinsolve 80 Carbon NMR.

FTIR Spectroscopy
Fourier transform infrared (FTIR) spectroscopy can be used to determine the molecular composition of specimens. The system at the SEC has a diamond ATR crystal for analysis of solids, powders and liquids. A TGA-IR accessory is also available for analysis of the gases evolved during TGA experiments.
More information on our Thermo Scientific Nicolet iS-50 FTIR.

DLS and Zeta Potential
To analyze particles, whether metal, ceramic, organic, or biological, the Zetasizer is available to characterize particle size from 2 nm to 2 µm via dynamic light scattering (DLS), and evaluate the stability of suspensions by zeta potential determination.
More information on our Malvern Zetasizer Pro.
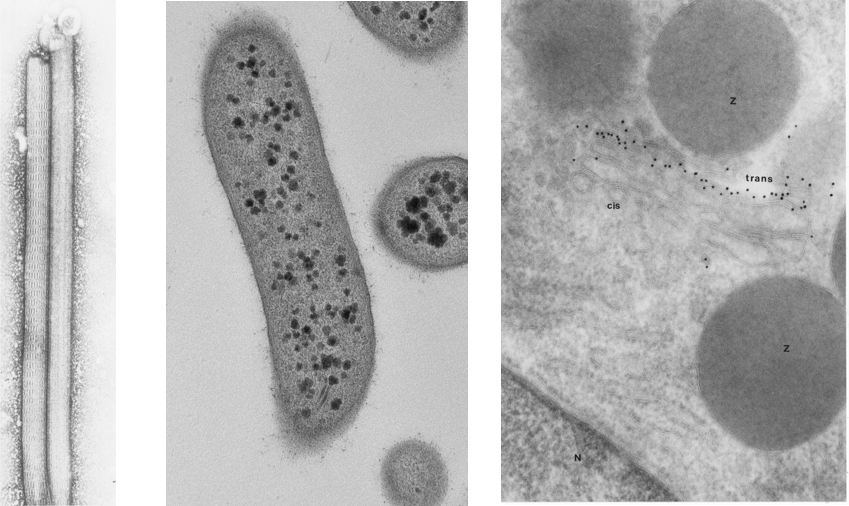
Biological Electron Microscopy
When preparing biological specimens for transmission electron microscopy , every step of the procedure affects the quality of the final images. Therefore, it is critical to process material according to prescribed protocols and understand what is happening in each step.
At CNS users can be trained on these sample preparation methods for a variety or sample types.
Negative staining is an established method for imaging the structure of proteins, single particles and small organisms, by contrasting a thin specimen with an optically opaque fluid. Routine morphology allows for specially prepared samples to be sectioned and high resolution of cellular infrastructure can be examined. Immuno cyto chemistry allows for the localization of proteins with in mildly fixed material. Users can be trained at CNS in these methodologies, or request service, or a combination of service and training depending on your research needs.
More information on our Hitachi 7800 TEM, Leica Ultramicrotome and Leica Cryo-Ultramicrotome.
Scanning Electron Microscopy
Scanning Electron Microscopy (SEM) is a versatile technique for studying sample surfaces. A field emission scanning electron microscope (FE-SEM) is available at the SEC for use on hard, soft and biological samples. This SEM has inlens and Everhart-Thornley detectors.
More information on our Zeiss Ultra 55 SEM.

Cryo-SEM
The technique of cryo-SEM is a powerful method to study bulk hydrated specimens. These samples are not readily suitable for conventional SEM analysis due to their incompatibility with vacuum conditions. In cryo-SEM, the specimen is rapidly frozen and then imaged at cryogenic temperatures; this allows the integrity of the specimen’s structure to be maintained for imaging without using processing techniques such as dehydration or chemical fixation.

Sample Preparation
A sample preparation area supports the electron microscopy work performed at the SEC. Sputter coating, carbon coating and plasma cleaning are available for preparation of SEM samples. A diamond wheel saw and polishing tool are helpful for making SEM and TEM samples from hard materials. Critical point drying can be used to dry soft and biological samples, and a glow discharge unit to prepare TEM grids. Sectioning can be performed using our room temperature and cryo ultramicrotome, while a fume hood is available for embedding and staining.

